
2024.05
22
Learn the Aroma Components of Sake - Ginjo Aroma
80% of the deliciousness that humans perceive is said to be due to aroma. There are over 100 aroma components in sake that determine the aromatic profiles in sake. Among them, an aroma known as ginjo-ka (ginjo aroma) has been scientifically recognized as having a relaxing effect in recent studies.
Reference: 世界初!日本酒の香りによるリラックス効果を月桂冠総合研究所が確認|Gekkeikan
In this article, I will explain, from a scientific perspective, the fruity aroma often found in ginjo category and the supporting flavors.
Ginjo-zukuri method: Polishing well and fermenting slowly
Ginjo-zukuri, is a name given to the brewing process that creates ginjo aromas and it defined as follows by the National Tax Agency:
Ginjo-zukuri refers to brewing with careful consideration. Traditionally, better polished rice is fermented slowly at low temperatures and brewed to have a distinctive aroma, ginjo-ka.
Although koji making styles and other factors may be related to the aroma, this article will focus only on the rice polishing ratio and fermentation temperature. First of all, one of the requirements for ginjo sake is a rice polishing ratio of 60% or less, but how does milling the rice relate to the components of the sake fermentation? The graph below shows the relationships between rice components and rice polishing.
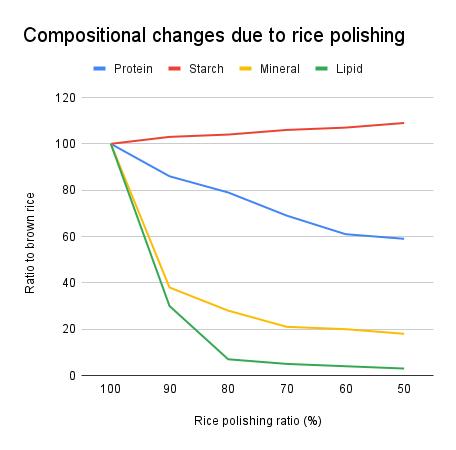
What is noteworthy in this graph is the reduction in lipid content. When the rice is polished to 60%, only about 5% of the lipid remains in the rice. This reduction in lipid affects the synthesis of ginjo aroma.
(*1) Based on "Properties of Sake Rice and Their Treatment (Part 1)" by S. Yoshizawa and Y. Ishikawa (1974), Journal of the Brewing Society of Japan, Vol. 69, No. 10.
Next is the temperature. The temperature of the non-ginjo sake is kept around 8-15℃ (46-59°F), but for ginjo sake, the temperature is controlled at 5-10℃ (41-50°F). This results in lower yeast activity and longer fermentation time for the non-ginjo sake. Fermentation at low temperatures alters the activity of yeast and enzymes, which in turn affects the ginjo aroma.
Let's take a look at the specific aroma components.
Apple-like aroma, ethyl caproate

Ethyl caproate, is one of two major ginjo aromas. This compound has an apple-like aroma: it is considered a relatively new aroma among ginjo aromas, but it can be found in many ginjo sake today. So how is the compound formed during the brewing process and why is it found in ginjo-style sake? :
How is ethyl caproate formed?
Ethyl caproate is synthesized by the reaction of caproic acid and ethanol.
Caproic acid + Ethanol → ethyl caproate
Caproic acid is a type of fat, and fat is often synthesized in yeast cells when the yeast consume sugar (when they are well nourished), just like our bodies. Caproic acid, when isolated, can smell like goat sweat. Not all of the caproic acid is changed, so it can remain in the drink a little, but the aroma of caproic acid can actually go well with fish roe such as sea urchin or salmon roe, or beef.
All yeasts synthesize lipids to survive, but this caproic acid is a lipid that only some yeasts can produce. Ordinary yeast can only produce fatty acids that are about three times larger than caproic acid, but yeasts such as Kyokai No. 1801, can produce not only large fatty acids, but also caproic acid, a relatively small fatty acid. High ethyl caproate-producing yeasts have this difference in fatty acid synthesis.
To synthesize a lot of ethyl caproate, you need more caproic acid and ethanol. Caproic acid is the key, since ethanol is abundant in moromi or fermentation mash. So, what comes next?
Ginjo-zukuri produces more caproic acid and reduces volatilization
Why do brewers use the ginjo-zukuri process? The answer is, quite simply, to increase the amount of caproic acid.
fermentation at lower temperatures results in a higher percentage of caproic acid in the fatty acids In addition to the larger fatty acids, I mentioned that high ethyl caproate-producing yeast has the ability to synthesize caproic acid as well. When fermented at low temperatures, the percentage of caproic acid among the fatty acids synthesized will be higher than when fermented at ordinary brewing temperatures.
By keeping the temperature low, volatilization is reduced Ethyl caproate is a substance that is soluble in ethanol and volatilizes in unrefined sake. Fermentation at low temperatures reduces volatilization in the mash and increases the amount of ethyl caproate that remains in the final sake.
Also, as is the case with many aroma components, by milling the rice and suppressing the aroma derived from the bran and fat on the outside, the desired aroma component is made to stand out relatively more.
Isoamyl acetate and the aroma of various related components
The other major ginjo aroma is isoamyl acetate. Let’s look at how it and other aromas are made.
While lipids were important for ethyl caproate, the key to isoamyl acetate is amino acids. Let's first look at how isoamyl acetate is synthesized. The pathway for isoamyl acetate is simply described as follows.
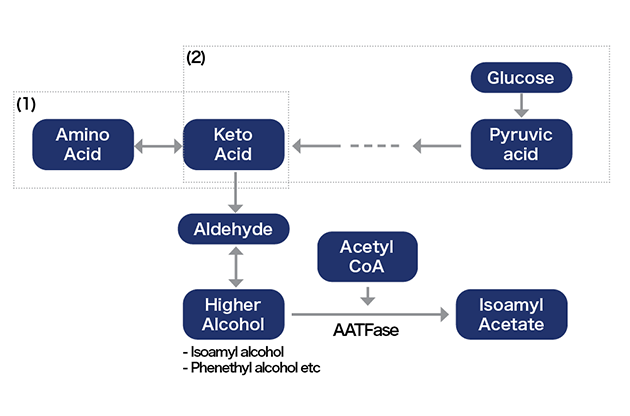
Flow from amino acids and sugars to isoamyl alcohol and isoamyl acetate
Thus, isoamyl acetate is synthesized primarily by altering substances in the pathways (1) that break down amino acids and (2) that are intermediate in the pathway that synthesizes amino acids.
For details,
[1-a] In the case of the amino acid degradation pathway: Amino acids become keto acids through deamination.
[1-b] In the case of the amino acid synthesis pathway: Pyruvic acid, which is formed from glucose, is metabolized to keto acid.
[2] Keto acids are decarboxylated to aldehydes.
[3] Aldehydes are reduced to higher alcohols
[4] Isoamyl alcohol, a type of higher alcohol, reacts with acetyl CoA (a substance produced by yeast to obtain energy) to form isoamyl acetate
Now that we have the big picture, let's take a look at the individual aromas.
Alcoholic aroma like permanent marker, isoamyl alcohol

It is called the key aroma (*2) of sake, and is the component that supports the aroma that is typical of sake. When people talk about "higher alcohol" in sake tasting, they are referring to isoamyl alcohol, which by itself is said to have an alcoholic aroma like that of permanent marker.
(*2) Key aroma [kicyo-ka]: Odor that remains in the remainder of the sake when it is left in air.
Isoamyl alcohol is synthesized by the breakdown of an amino acid (leucine), as shown in the figure above.
Incidentally, the aldehyde used in the synthesis of isoamyl alcohol is called isovaleraldehyde and is a component of old-fashioned Asian incense. Note that if the raw sake is left at room temperature, the enzymes will change the isoamyl alcohol.
Banana-like aroma, isoamyl acetate

Isoamyl acetate has a banana-like aroma and, as mentioned earlier, is parallel to ethyl caproate as mentioned earlier. It is produced when isoamyl alcohol and acetyl CoA are combined by the enzyme AATFase.
Isoamyl alcohol + Acetyl CoA → Isoamyl acetate
The key enzyme in this reaction is AATFase. This enzyme is a little unstable and must be fermented at low temperatures in order for it to work stably. In addition, the function of AATFase is inhibited by the presence of unsaturated fatty acids. Unsaturated fatty acids are produced when the fat that is abundant on the outside of rice is broken down by the enzymes of koji. Therefore, if the rice is polished and the fat is removed, the function is stabilized and isoamyl acetate is synthesized in a stable manner. This is why ginjo-style sake is made.
Also, unlike the caproic acid mentioned earlier, amino acids are metabolized in the same way by all yeasts. For this reason, you can expect to find isoamyl acetate in sake made with ginjo-style brewing, albeit in varying concentrations.
In order to synthesize more isoamyl acetate, yeast that synthesizes more isoamyl alcohol has also been studied and developed. This yeast has a change in one of the enzymes that synthesizes leucine and has the ability to synthesize more leucine and also more isoamyl alcohol, resulting in an increase in the amount of isoamyl acetate.
Glue-like aroma, ethyl acetate

Here is a description of the aromatic components of sake, which have a little to do with this pathway.
First is ethyl acetate. Ethyl acetate is synthesized when acetyl CoA and ethanol, which were mentioned earlier, are combined by AATFase. Both of these substances are abundant in sake unrefined sake, and can be said to be the general aroma of sake.
In sake tasting, excessive one is described as having a “Cemedine" odor (the smell of nail polish or glue), but it is actually an aroma component found in ripe bananas and melons, and is thought to affect the fruity aroma of sake as well.
Rose-like aroma, phenethyl alcohol

Phenethyl alcohol is synthesized by the same synthetic pathway as isoamyl alcohol, but with a different target amino acid: phenylalanine, which is synthesized by the breakdown of the amino acid phenylalanine.
It may not be a familiar aroma, but it is considered to be the key aroma of sake, and research and development is underway to develop yeast that can produce more of this aroma. The fragrance of interest is a sweet, floral scent like that of roses, and is also used in cosmetic fragrances.
Other fruit-like aroma
Muscat and lychee-like aroma, 4MMP

It is a new aroma for sake, and is currently being considered for inclusion in the "quality evaluation terminology" being created by the National Research Institute of Brewing. The aroma is reminiscent of muscat or lychee, and is the characteristic aroma of a type of hop used in white wine Sauvignon Blanc and craft beer.
It is said that sake tends to produce 4MMP aroma when brewed with low glutelin rice, which has low glutelin (easily digestible protein) content, and when yeast decomposes certain substances. It is believed that 4MMP will become a major aroma in sake in the future, and if this component can be controlled, the range of aromas will expand even further.
In addition to the aromas mentioned above, various other aromas, such as green apple and peach, are mixed in a complex way to create the fruit-like aroma of sake.
Summary
The aromas listed here are just a handful of the more than 100 aromatic components of sake. Aromas involve complex compounds, and I believe that a deeper understanding of some of the major aromas will lead to new ways to enjoy sake. When you drink sake and smell the aroma, try to think about how the sake was brewed from a scientific point of view.
References
- 選んで育てて!香味豊かなオリジナル清酒酵母 [Choose and Grow! Original aromatic sake yeast] (Gekkeikan Website)
- Tips for B・F・D 連載第26回 清酒のにおい・かおりとその由来(その1) [Tips for B-F-D Column #26 Sake’s aroma and their origins] Hitoshi Utsunomiya (Kita Sangyo Website)
- 生物工学会誌 89巻12号「清酒酵母の香気生成の研究」 [Journal of Bioscience and Bioengineering 89-12, Study of aroma production by sake yeast] Hiroko Tsutsumi (2011)
- Journal of the Brewing Society of Japan 88-2「カプロン酸エチル高生産酵母」 [Yeasts that produce high levels of ethyl caproate] Eiji Ichikawa (1993)
- Journal of the Brewing Society of Japan 84-11「酵母による香気生成」 [Aroma production by yeast] Osamu Akita (1989)
- Journal of the Brewing Society of Japan 98-3 「β-フェネチルアルコール高生産清酒酵母の分離とその生成機構」 [Isolation and production mechanism of β-Phenethyl alcohol high-producing sake yeast] Kazuyoshi Koganemaru et al. (2003)
- Onkochishin Vol.56 (Akita Konno) マスカットやライチ、柑橘の香りがするお酒 : 清酒の新しい香り 4-mercapto-4-methylpentan-2-one(4MMP) [Sake with aromas of muscat, lychee and citrus: a new aroma of sake 4-mercapto-4-methylpentan-2-one(4MMP)] Sachiko Iizuka (2019)
- Journal of the Brewing Society of Japan 82-3 「清酒もろみの香気成分の保留」 [Withholding of aromatic components of moromi mash of sake] Kiyoshi Ito (1987)
Pickup Articles
2019.01.18
2019.01.25
Trending Articles
Popular Articles
Recent Articles













